Students should refer to Calorimetry ICSE Class 10 Physics notes provided below designed based on the latest syllabus and examination pattern issued by ICSE. These revision notes are really useful and will help you to learn all the important and difficult topics. These notes will also be very useful if you use them to revise just before your Physics Exams. Refer to more ICSE Class 10 Physics Notes for better preparation.
ICSE Class 10 Physics Calorimetry Revision Notes
Students can refer to the quick revision notes prepared for Chapter Calorimetry in Class 10 ICSE. These notes will be really helpful for the students giving the Physics exam in ICSE Class 10. Our teachers have prepared these concept notes based on the latest ICSE syllabus and ICSE books issued for the current academic year. Please refer to Chapter wise notes for ICSE Class 10 Physics provided on our website.
Calorimetry ICSE Class 10 Physics
Temperature
Heat is a form of energy that can be converted into other forms of energy (like mechanical energy, electrical energy, etc.) and vice versa. We use heat for various purposes such as cooking.
One hot day, on returning from school, Ravi touched the iron gate leading to his house. He found it to be extremely hot. Later, he touched other things made of iron present inside his house and found that they were not hot. Then, he touched other substances (not made of iron) present in his house to determine whether they were hot or cold. He
listed his observations as in the table given below.
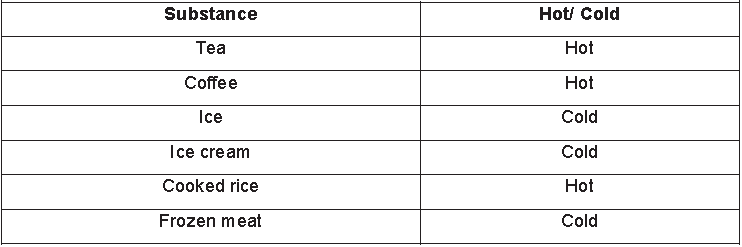
Try to make a table, listing some other substances present in your house and classify them as hot or cold. But how do you decide whether a substance is hot or cold? Can you always tell whether a substance is hot or cold simply by touching it? The following activity will help you understand the concept better.
Activity:
Take three containers and label them as A, B and C. Take hot water in container A, and cold water in container C. In container B, mix hot and cold water in equal amounts. Now, place your left hand in container A and right hand in container C for two minutes. Then, dip both your hands in container B.

What can you say about the water present in the three containers? Note down your observations in the table given below.

What is your observation for container B? Is the water in container B hot or cold?
When you dip your hands in container B, your left hand will tell you that the water is cold, while your right hand will tell you that the water is hot. Thus, you will not be able to distinguish whether the water present in container B is hot or cold.
From this activity, we can conclude that we cannot decide whether a substance is hot or cold just by touching it. Thus, we need something more reliable than our sense of touch to decide whether a substance is hot or cold.
Is Heat Measurable?
The measure used for detecting the degree of hotness of a substance is called temperature.

Think and tell- How do you come to know that your mother is cooking something special for you in the kitchen?
Yes, you can smell it from your room. But do you know why you are able to do so?
Every substance is made up of matter and matter is made up of atoms and molecules. These atoms and molecules possess kinetic energy and are constantly in motion. Also, heat is a form of energy. So, if we heat any substance, we are providing energy to it. This added energy increases the kinetic energy of the molecules and thus, increases the temperature of the substance.
The total kinetic energy of all the molecules in a substance is called heat and the increase in average kinetic energy of molecules of the substance is called temperature. The S.I. unit of heat is Joule (J) and the S.I. unit of temperature is Kelvin (K).
Heat depends upon the mass of the substance, while the temperature does not.
Let us differentiate between heat and temperature.

Relation between degree Celsius and Kelvin
T K = 273 + t °C
Hence, by adding 273 to the degree Celsius temperature we get Kelvin temperature.
t°C = T K − 273
If 273 is subtracted from Kelvin temperature, we get degree Celsius temperature.
Specific Heat and Specific Heat Capacity
Take the same amount of water in two similar beakers. Put a thermometer in each beaker, and heat the beakers. Heat one beaker for 5 minutes and the other for 10 minutes. What do you observe?
You will observe that the water in the beaker that is heated for the longer time becomes hotter than the other beaker. The thermometer in this beaker shows a higher temperature. Why does this happen?
The answer is very simple—the longer the time we heat water, the hotter it becomes. So, we can say the rise in the temperature of a body is directly proportional to the heat energy contained in the body.
Let us do an experiment to explore more properties of heat.

As shown in the figure, take two beakers: one with 100 mL of water and the other with 200 mL of water. Now, start heating the beakers, and heat them till the temperatures in both the beakers become the same. Do they require the same time?
You will observe that the time taken to raise the temperature of water by 30°C in the 200-mL beaker is double the time taken to raise the temperature of water (by the same measure) in the 100-mL beaker. Perform this experiment a few more times by raising the temperature to different levels.
Every time you will get the same result.
Hence, we can conclude that the amount of heat absorbed by a body to raise its temperature is directly proportional to the mass of the body. The more the mass of a body, the more is the heat required by it to raise its temperature.
Again, we take the same two beakers. Fill the beaker containing 200 mL of water with 100 mL of oil instead.

Heat the two beakers for the same amount of time. You will observe that the temperature of oil increases more than that of water for the same amount of heat energy supplied. Thus, it can be concluded that water needs more heat energy than oil to be raised to the same degree of temperature.
Hence, it can be concluded that heat energy contained in a body is directly related to the chemical composition of the body.
Therefore, from all the experiments we can conclude that
• When the mass (m) is constant, the rise in the temperature (Δθr ) of a body is directly proportional to the heat energy (H) supplied to it, i.e., H ∝Δθr (mis constant)
• When the change in temperature is constant, the heat energy (H) absorbed by an object is directly proportional to the mass (m) of the object, i.e.,H∝ m(Δθris constant)
• When both mass (m) and temperature change (Δθr ) are constant, the heat energy (H) absorbed by an object depends on its chemical composition.
• Therefore, we can say that
H∝ mΔθr(Both m andΔθr are variable)
∴ H = mCΔθr
Where, C is a proportionality constant, called specific heat capacity. This depends on the nature of the substance.
Thus,
Heat absorbed = Mass of the body × Specific heat capacity × Rise in temperature Units of Specific heat capacity

In the CGS system, the unit of heat is dyne centimeter (dyn-cm); that of mass is gram (g), and that of temperature is °C.
Therefore, the unit of specific heat capacity in the CGS system is (dyn-cm g−1 °C−1) In the SI system, the unit of heat is joule (J); that of mass is kilogram (kg), and that of temperature is Kelvin (K).
Therefore, the unit of specific heat capacity in the SI system J kg−1 K−1 Water has the highest specific heat capacity of 4.2 J/g °C. In other words, every 1 g of water will absorb 4.2 J of energy when its temperature rises by 1°C.
Measurement of Specific Heat
Measurement of specific heat of an object includes two components: mixing method and calorimeter. In this method, a hot solid object is put in the water in a calorimeter. As soon as the hot object is placed in water, exchange of heat occurs between the hot object and water in the calorimeter. This exchange of heat continues till the temperatures of the solid object, water and the calorimeter become equal. So, we can say that
Heat lost by solid object = Heat gained by water in calorimeter + Heat gained by the
calorimeter …..(1)
The following equation will give us an idea about how to calulate the varoius components which are present in the above equation.
Heat lost by the solid object (Q) = Mass of the solid object ×× Its specific
heat ×× Decrease in its temperature …..(2)
Heat gained by the water (Q1) = Mass of the water ×× Its specific heat ×× Increase in its
temperature …..(3)
Heat gained by the calorimeter (Q2) = Mass of the calorimeter ×× Its specific
heat ×× Increase in its temperature …..(4)
Using equation 2, 3 and 4, we can rewrite equation 1 as:
Q = Q2 + Q1

Relationship between heat capacity and specific heat capacity
Heat capacity, C” = Mass, m X Specific Heat Capacity, C”
Calorimeter
Principle of Calorimetry
Calorimetry means measurement of heat. When a body at higher temperature is brought in contact with a body at lower temperature, the heat lost by the former is equal to the heat gained by the latter (no heat should escape to the surroundings).
Calorimeter
A device used for heat measurement is called a calorimeter.
Construction of a Calorimeter
• It consists of a metallic vessel and stirrers. They are made of copper or aluminium.
• The vessel is then kept inside a wooden jacket which contains heat-insulating materials.
• The outer wooden jacket acts as a heat shield, and reduces the heat loss from the inner vessel.
• The outer jacket has an opening through which a mercury thermometer is inserted into the calorimeter.
Determination of Specific Heat by a Calorimeter
• Consider a calorimeter of known water equivalent containing water.
• Note the initial temperatures of the water and the calorimeter.
• Heat the substance whose specific heat is to be determined to a particular temperature.
• Put this substance in the calorimeter, and stir the mixture.
• The substance at higher temperature will lose heat, which will in turn be gained by the water and the calorimeter.
• Stir the mixture and note the constant temperature.
• Weigh the mixture to find the mass of the added substance.
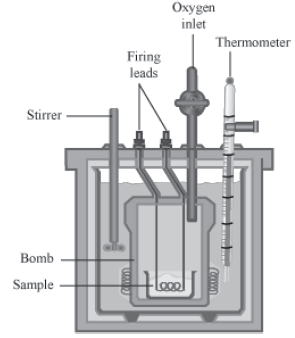
Consider,
m1 = Mass of water
t1 = Initial temperature of the water and the calorimeter
w = Water equivalent of the calorimeter and the stirrer
m2 = Mass of the substance
s = Specific heat of the substance
t2 = Temperature of the substance
t = Common temperature of the mixture
Rise in the temperature of the water and the calorimeter = (t − t1)
Fall in the temperature of the substance = (t2 − t)
Heat gained by the water and the calorimeter = (m1 + w) (t − t1)
Heat lost by the substance = s. m2 (t2 − t)
According to calorimetry principle,
(m1 + w) (t − t1) = s.m2 (t2 − t)

Note
Rise in the temperature of a body may not be equal to the fall in the temperature of another body.
Change of State
We know that matter exists in three different physical states − solid, liquid, and gas. The states of matter are interchangeable i.e., a substance can be changed from its solid state to its liquid state and vice-versa. Transitions from the liquid state to the gaseous state and vice-versa are also possible. However, some substances directly change into
their gaseous state from their solid state and vice-versa.
Water exists in all three states (i.e., solid, liquid, and gaseous) as ice, water, and water vapour respectively. These states are interchangeable (see the given figure).
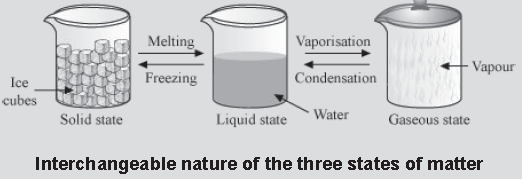
Let us now explore how matter changes its form.
Temperature is a major factor responsible for causing a change in the various states of matter. The changes in the states of matter with increasing or decreasing temperatures can be shown in the form of the following figure.
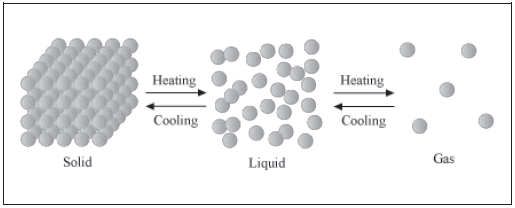
Effect of temperature on the states of matter
On heating a solid substance, the kinetic energy of its constituent particles increases. As a result, the particles start vibrating with greater speed. This extra energy helps the particles to overcome the inter-particle force of attraction. Soon, they leave their positions and start moving more freely. Consequently, the substance melts. The change of solid state into liquid state is known as melting. The minimum temperature at which a substance starts melting and becomes a liquid at atmospheric pressure is known as its melting point.
Heating curve of ice during melting
To obtain this curve, we plot a graph showing variation in temperature of ice at various instant of time when a test-tube containing ice is placed on a burner. The graph is plotted until the temperature of water (obtained after melting of ice) reaches upto to 30 oC. The reading is taken by the thermometer gently inserted into the ice such that it does not touch the walls of the tube.

From the graph it clear that temperature of ice in the part AB remains equal to 0 oC till the whole ice melts. The constant temperature at which the whole ice melts is known as the melting point of ice.
Effect of pressure on melting point
• By increasing the pressure, the melting point decreases for the substances which contract on melting . For instance ice.
• By increasing the pressure, the melting point increases of the substances which expand on melting. For instance lead, wax etc..
Latent heat
The heat energy absorbed or liberated during the change of state of a matter and which does not actually take part in rising or falling of the temperature of the matter is called latent heat of the matter.
Now, this latent heat when expressed for unit mass of a matter is called the specific latent heat and it is denoted by letter L.
Specific Latent Heat

Hence, the specific latent heat of a state is the quantity of heat energy absorbed or liberated by the unit mass of the substance for the change in its state at a constant temperature. S.I. unit of specific latent heat is J kg-1.
Do you know!
It has been observed that temperature remains constant till all solid melts into liquid. The heat supplied is used for changing a solid state into its liquid state by overcoming the particle-particle attraction force. The solid substance absorbs heat energy without showing any rise in temperature. This heat energy is called latent heat. The amount of
heat required to convert a unit mass of a solid into its liquid state without a change in temperature (i.e., at its melting point) is called the latent heat of fusion.
For ice, the latent heat of fusion is 334 kJ kg-1. This means that 334 kJ of heat is required to convert 1 kg of ice at 0ºC into 1 kg of water at 0ºC. Conversely, 334 kJ of heat is released when 1 kg of water freezes at 0ºC to give 1 kg of ice at 0ºC.
Natural consequences of high specific latent heat of fusion of ice
• Snow on high mountains does not melt all at once
• In extremely cold conditions, water in the lakes and ponds does not freeze all at once
• Pieces of ice can cool a drink more quickly than the ice-cold water at 0 oC
• Surrounding of a frozen lake becomes extremely cold if it starts melting
If heating is continued, then the kinetic energy of liquid particles increases further. This increases the velocity of the particles, and at a certain temperature, they obtain enough energy to break free from the force of attraction of the other particles. At this point, the liquid starts boiling i.e., starts changing into its gaseous state. This temperature is
known as the boiling point of the liquid.
Heating curve for water
Take some water at 20 oC (room temperature) in a flask and suspend a thermometer in it. Now heat the flask on a burner and observe the temperature after every half an hour till the water starts boiling and vapours are present in the flask. We will observe that the bubbles are formed throughout the water showing that boiling occurs throughout the
volume of water.
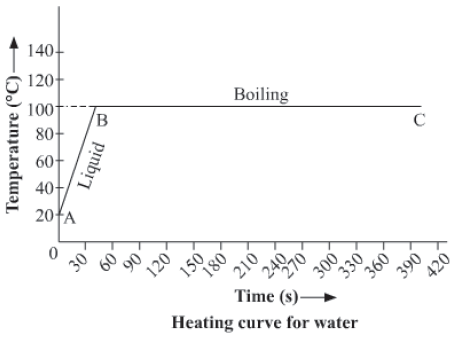
By plotting the graph of temperature against time, we see that the temperature of water rises continuously in the curve AB where it is in liquid state and this is because of the absorption of heat by water continuously. Now, at point B the boiling starts and the temperature does not increase further which is shown by the curve BC even thought there is also continuous absorption of heat energy. This straight line BC parallel to time axis represents the boiling of water and the point B corresponds to boiling point of water.
Effect of pressure on boiling point
• The boiling point of liquid increases with increase in pressure and decreases with decrease in pressure
The latent heat of vaporisation is the amount of heat required to convert a unit mass of liquid into its vapour state, without a change in its temperature. Latent heat of vaporisation of water is 2,260 kJ kg-1. This means that 2,260 kJ of heat must be provided to convert 1 kg of water at 100ºC into 1 kg of vapour at 100ºC. Conversely, 2260 kJ of heat is released when 1 kg of vapour condenses at 100ºC to give 1 kg of water at 100ºC.
Conversely, when gas is cooled beyond its boiling point, it changes to its liquid state. This process is called condensation. When a liquid is cooled beyond its freezing point, it solidifies. This process is called freezing. The freezing point and boiling point of water are 0°C and 100°C respectively.
When a solid directly changes into its gaseous state on heating, the process is called sublimation. The reverse process is called deposition. Sublimation can be observed with the help of the following activity.
Have you ever heard about the term regelation?
Regelation refers to the phenomenon in which the ice converts to liquid due to applied pressure and when the pressure is removed, then it re-converts into ice. The reason behind this change is that the increased pressure lowers the melting point of ice below 0°C, which means that at 0°C, the ice gets converted into water. As the pressure is
removed, the melting point is restored to 0°C, which results in water getting converted into ice again.







